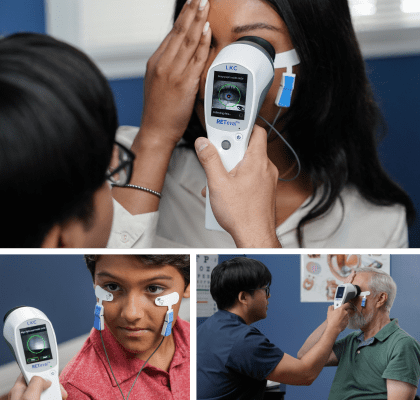
Electroretinography isn’t what it used to be. FDA-cleared and made in the US, the RETeval® offers a portable, handheld solution that is comfortable for patients, easy for clinicians, and provides an efficient, objective functional assessment of retinal health.
COMFORTABLE FOR PATIENTS
- Patented Sensor Strip adhesive skin electrodes save time & reduce hassle
- Flexible Sensor Strip material is gentle on skin with no corneal contact required
- Appropriate for any age without sedation
- Dilation not required
Validated by 200+ Peer-Reviewed Research Studies
Since its launch in 2015, more than 3,000 devices have been installed worldwide in optometry, ophthalmology, retina specialty, and research settings. LKC Technologies has been an industry leader in functional assessments for more than 45 years, and the RETeval is backed by science.
How It Works
- The RETeval device starts flashing light into the patient’s eye.
- The retina responds to the flashes by generating small electrical signals that travel through the facial structure to the Sensor Strip.
- The Sensor Strip detects the electrical signals and compares the results to the age-adjusted reference database.
